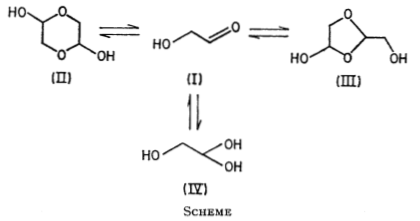
In 1971, Collins and George reported the equilibrium of glycolaldehyde in water by using NMR (Nuclear Magnetic Resonance).
IBM RXN for chemistry is a free AI web for predicting reactions. I plan to test IBM RXN with the isomerization of glycolaldehyde to see if it can get the right results.
Firstly, glycolaldehyde and H2O are input.
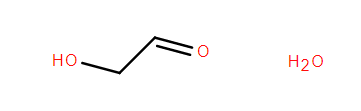
We get the answer quickly but with low confidence.

The AI system seems not to be aware of the equilibrium between glycolaldehyde monomer and dimer.
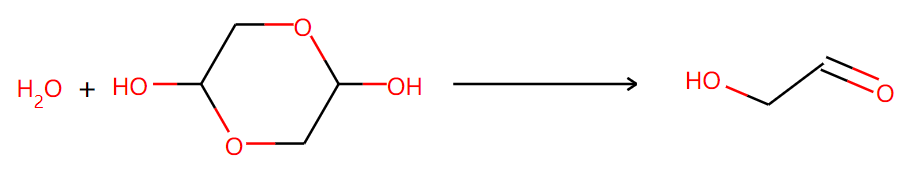
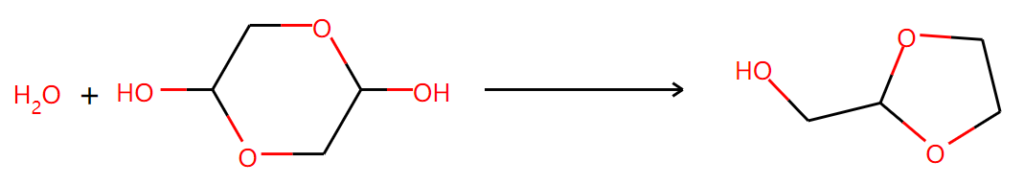
Then, what will happen if the six-member ring dimer is input? It is needed to point out that the glycolaldehyde exists as a dimer in the solid phase. AI gives 5 potential reactions with low confidence. The results are partly right.
Two of them are right:
Two of them are wrong obviously:
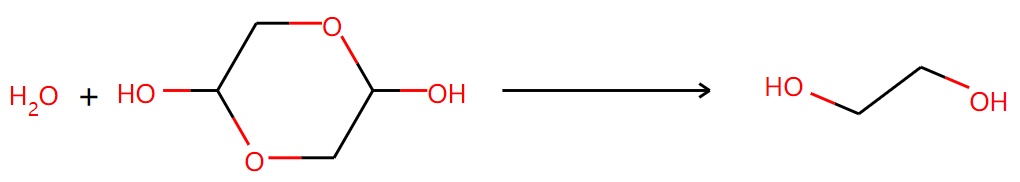
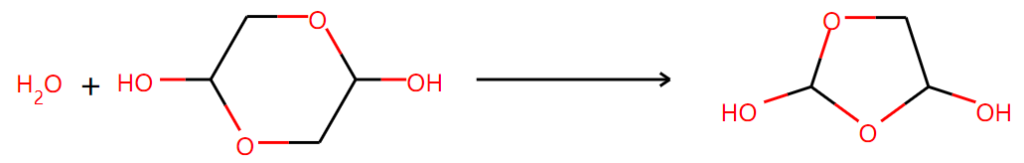
Another one is not supported by NMR results even though it seems to be reasonable:
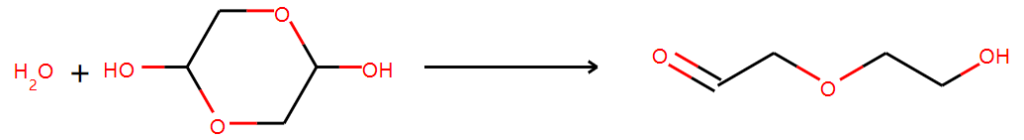
References:
Collins G C S, George W O. Nuclear magnetic resonance spectra of glycolaldehyde[J]. Journal of the Chemical Society B: Physical Organic, 1971: 1352-1355.